 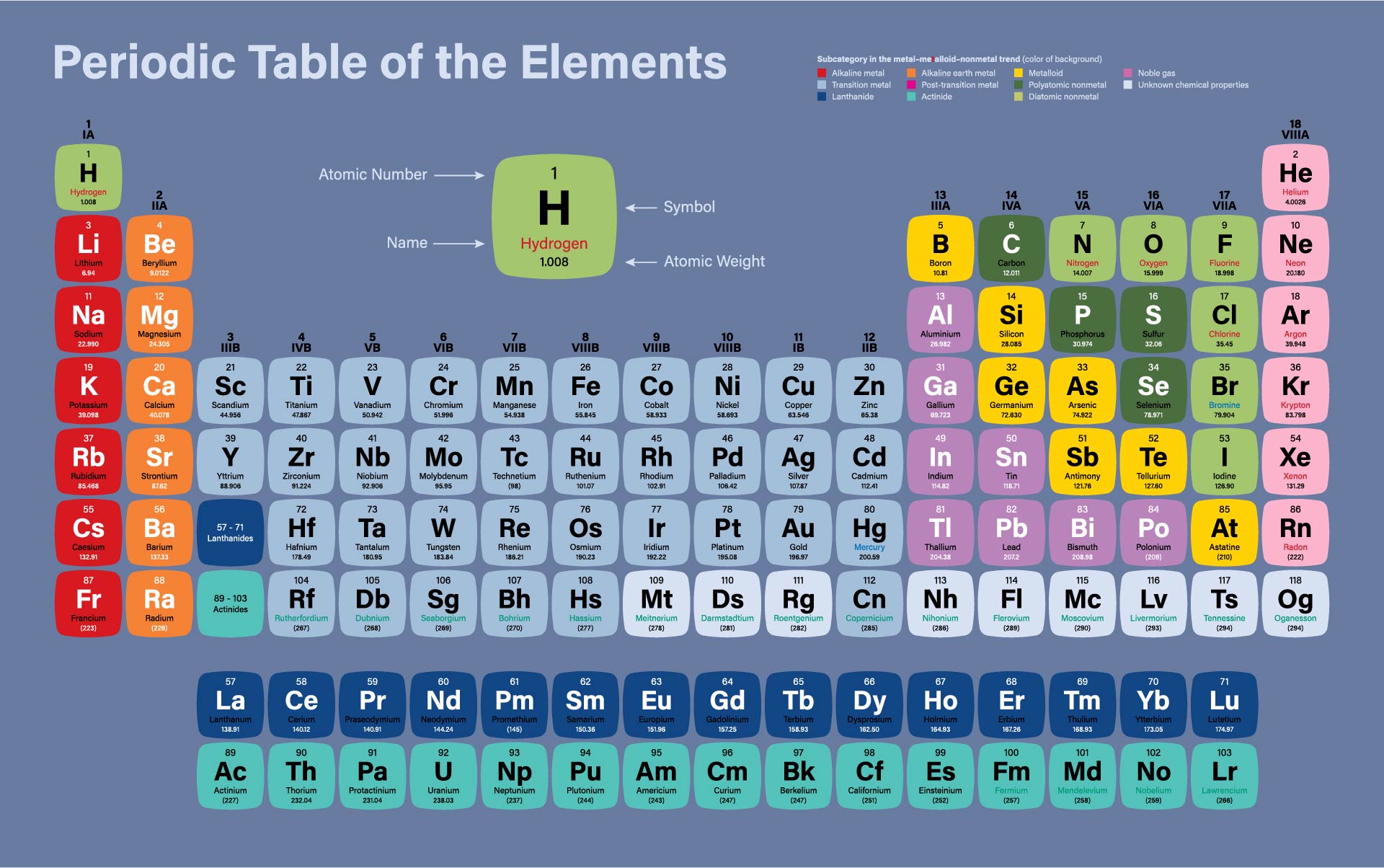
Protons are relatively heavy particles with a charge of 1+ and a mass of 1.0073 amu. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. The number of electrons in a neutral atom is equal to the number of protons. For example, if the symbol is 14 6 C, the number 6 is. /ColorPeriodicTableEC-58b5c7fa3df78cdcd8bbb56f.png)
Usually, both the mass number and atomic number are given in an isotope symbol. Look for the symbol C on the periodic table to get the atomic number. For example, if the symbol is 14 C, you know the element symbol is C. An amu is defined as exactly 1/12 1 / 12 of the mass of a carbon-12 atom and is equal to 1.6605 × × 10 24 g. The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. Find the atomic number from the isotope symbol the same way. This was mainly because the idea of atoms being made up of smaller sub-atomic particles (protons, neutrons and. Historically, however, relative atomic masses were used by scientists trying to organise the elements. The observed properties of gallium and germanium matched those of eka-aluminum and eka-silicon so well that once they were discovered, Mendeleev’s periodic table rapidly gained acceptance. The mass of one atom is usually expressed in atomic mass units (amu), which is referred to as the atomic mass. The modern periodic table lists the elements in order of increasing atomic number (the number of protons in the nucleus of an atom). Two of the blanks Mendeleev had left in his original table were below aluminum and silicon, awaiting the discovery of two as-yet-unknown elements, eka-aluminum and eka-silicon (from the Sanskrit eka, meaning “one,” as in “one beyond aluminum”). Periodic Table With Atomic Mass: Electrode Potential: Valency Of First 20 Elements: Chlorine Formula: Comments. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.\)). The atomic number of an atom is equivalent to the total number of electrons present in a neutral atom or the total number of protons present in the nucleus of an atom. Please note that the elements do not show their natural relation towards each other as in the Periodic system. 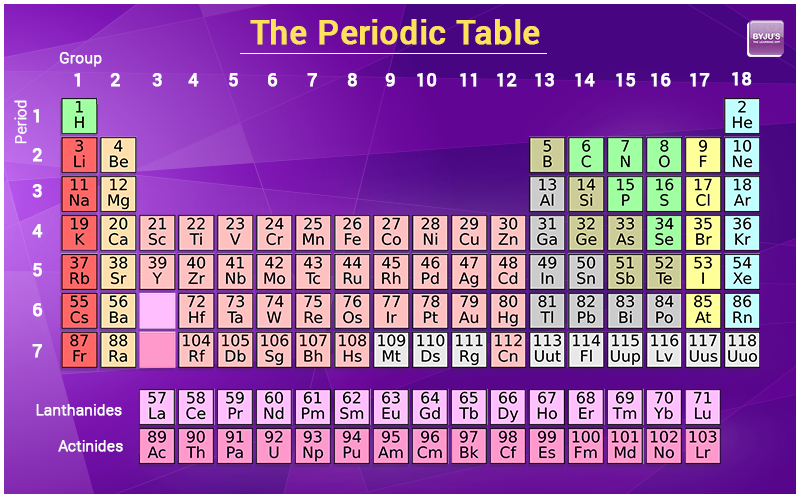
The first chemical element is Hydrogen and the last is Ununoctium. Where more than one isotope exists, the value given is the abundance weighted average. It is the same as the number of protons that the atom of. This is approximately the sum of the number of protons and neutrons in the nucleus. We remember from our school chemistry course that every element has its own specific atomic number. Relative atomic mass The mass of an atom relative to that of carbon-12. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic number. In this example the atomic number is 17 and the mass number is 35. Density is the mass of a substance that would fill 1 cm 3 at room temperature. This list contains the 118 elements of chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
